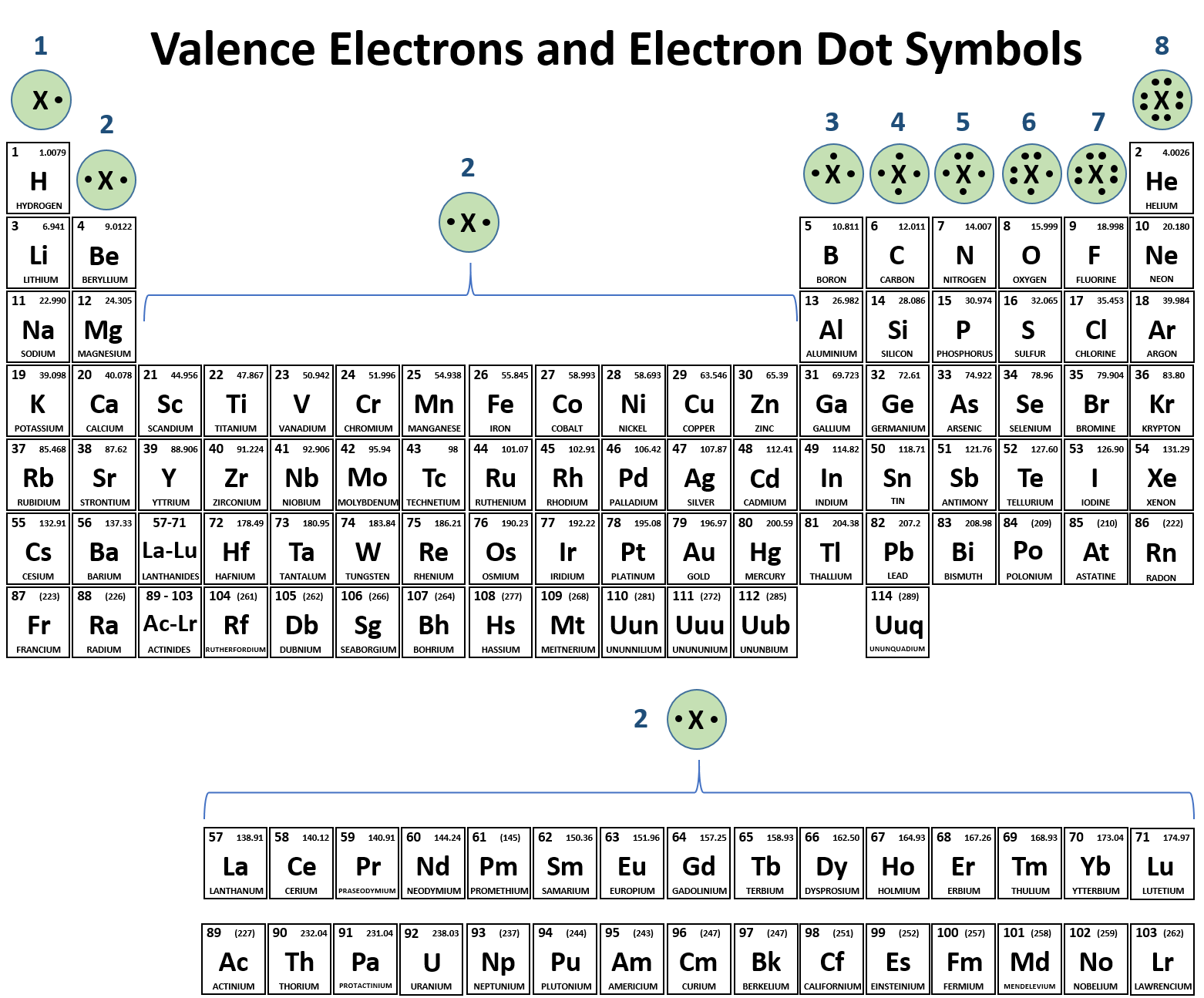
Which Elements Form Ionic Bonds
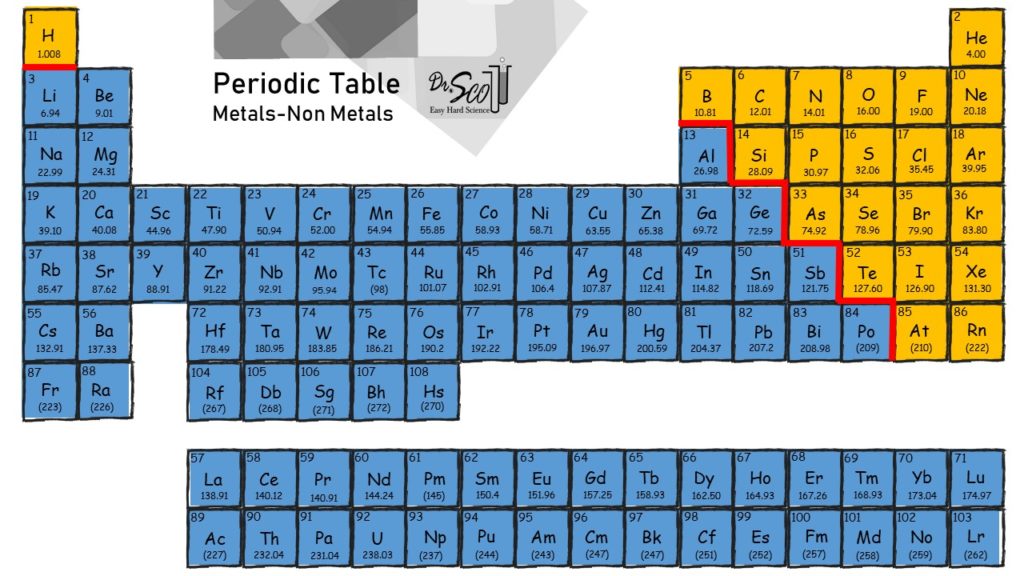
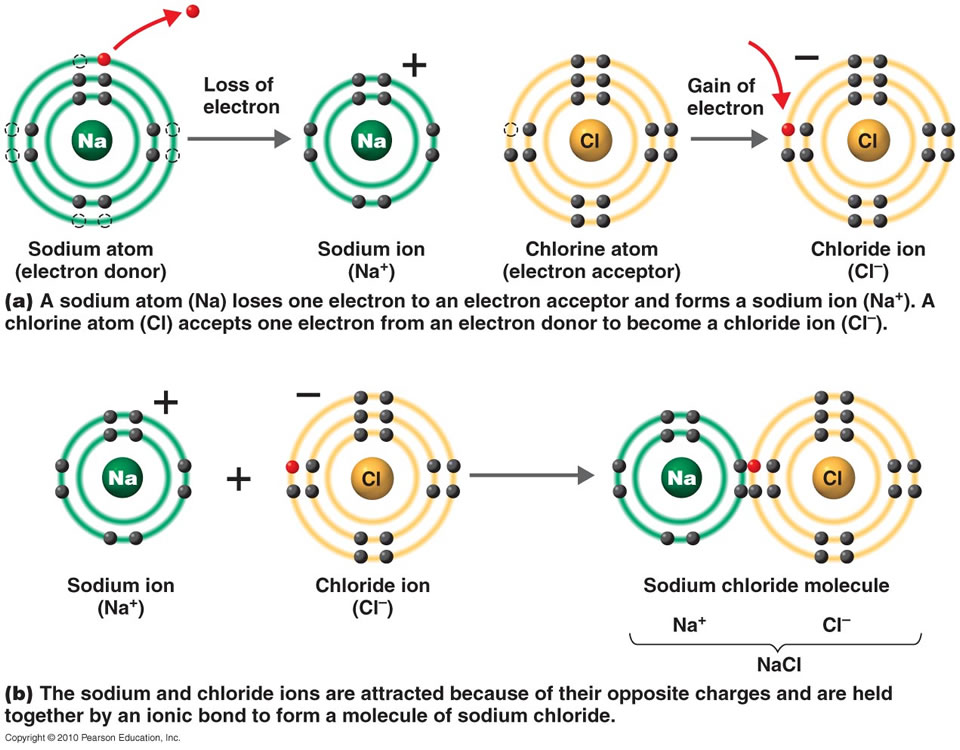
Which Elements Form Ionic Bonds - 3) last example, mg and cl. Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. Web between which type of elements do ionic bonds occur, and how do electrons act within the bond? One has been shown for you. Web barium (ba) phosphorus (p) radon (rn) lithium (li) Which of the following elements is likely to form ionic bonds? Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. Metals and nonmetals, electrons transferred. Metals and metals, electrons freely moving. One that tends to lose electrons and one that tends to gain them suppose an oxygen atom gains two electrons to become an oxygen ion.
Ionic bonds covalent bonds nacl. 1) for example, consider na and cl. Which of the following elements is likely to form ionic bonds? In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. Web use the periodic table to describe which elements form ionic bonds and which elements form covalent bonds. Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. Web between which type of elements do ionic bonds occur, and how do electrons act within the bond? Web which elements form ionic bonds? One that tends to lose electrons and one that tends to gain them suppose an oxygen atom gains two electrons to become an oxygen ion. Web ionic bonding is the complete transfer of valence electron(s) between atoms.
One has been shown for you. Web between which type of elements do ionic bonds occur, and how do electrons act within the bond? Web study with quizlet and memorize flashcards containing terms like review the discussion of electronegativity and the discussion of ionic bonds in section 2.2. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. Name at least five ionic bonds and five covalent bonds and enter them in the table below. Web ionic bonding is the complete transfer of valence electron (s) between atoms and is a type of chemical bond that generates two oppositely charged ions. Web ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Web which elements tend to form ionic bonds? Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. Which of the following elements is likely to form ionic bonds?
Examples of Ionic Bonds and Compounds
Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. Web which elements tend to form ionic bonds? In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. (b) list five.
Ionic Bond Definition Easy Hard Science
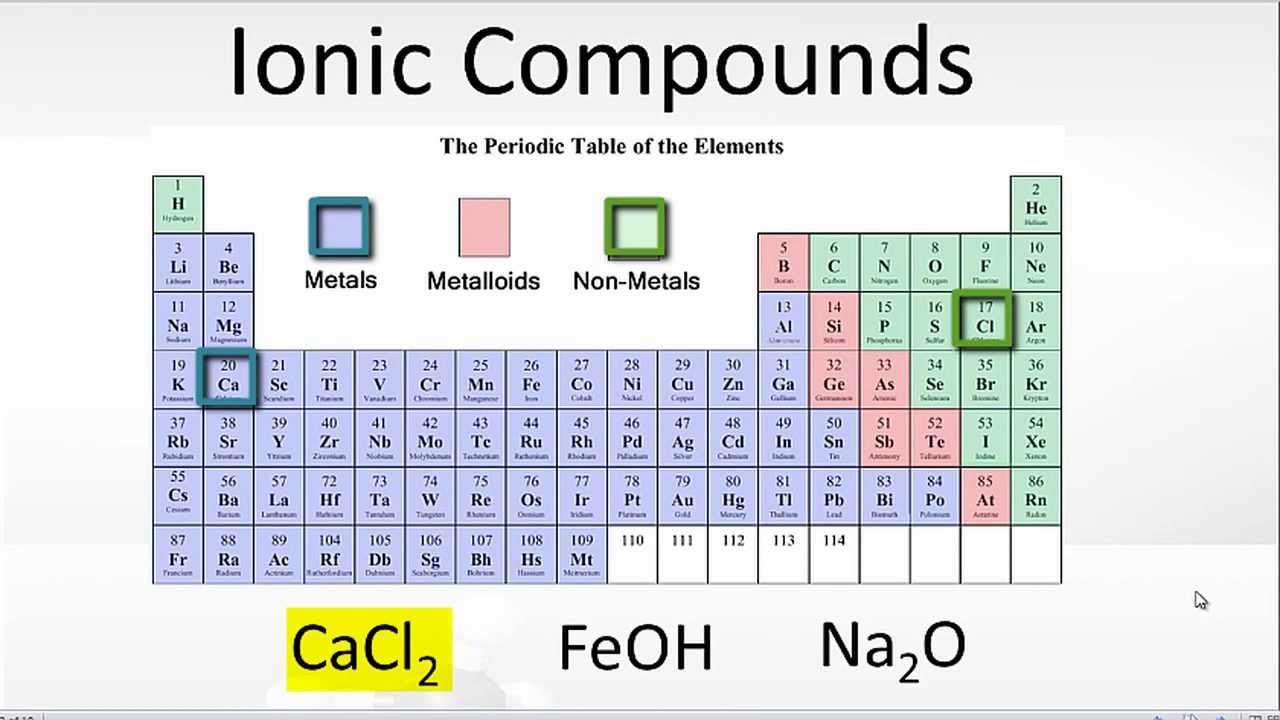
(b) list five examples of compounds with ionic bonds. Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. Web ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. One has been shown.
Examples of Ionic Bonding YouTube
1) for example, consider na and cl. Web ionic bonding is the complete transfer of valence electron(s) between atoms. The atom that loses the electrons becomes a positively charged ion (. Ionic bonds covalent bonds nacl. (b) list five examples of compounds with ionic bonds.
Ionic Bond Definition, Types, Properties & Examples
Web between which type of elements do ionic bonds occur, and how do electrons act within the bond? In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. 1) for example, consider na and cl. Web study with quizlet and memorize flashcards containing terms like.
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
The atom that loses the electrons becomes a positively charged ion (. Web ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. Web study with quizlet and memorize flashcards.
How Does An Ionic Bond Form Between Sodium And Chlorine slideshare
2) another example, magnesium and oxygen. One that tends to lose electrons and one that tends to gain them suppose an oxygen atom gains two electrons to become an oxygen ion. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. Name at least five.
Ionic Properties
Web which elements tend to form ionic bonds? Web study with quizlet and memorize flashcards containing terms like review the discussion of electronegativity and the discussion of ionic bonds in section 2.2. Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. Metals and nonmetals, electrons.
Ionic Bond Definition, Types, Properties & Examples
2) another example, magnesium and oxygen. Web which elements tend to form ionic bonds? In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. Web which elements form ionic bonds? Web between which type of elements do ionic bonds occur, and how do electrons act.
Ionic bonding Wikipedia
Web ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. It is a type of chemical bond that generates two oppositely charged ions. 3) last example, mg and cl. Metals and metals, electrons freely moving. One has been shown for you.
CH150 Chapter 3 Ions and Ionic Compounds Chemistry
Metals and nonmetals, electrons transferred. Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. One has been shown for you. Web use the periodic table to describe which elements form ionic bonds and which elements form covalent bonds. It is a type of chemical bond.
Ionic Bonds Covalent Bonds Nacl.
2) another example, magnesium and oxygen. Web study with quizlet and memorize flashcards containing terms like review the discussion of electronegativity and the discussion of ionic bonds in section 2.2. Which of the following elements is likely to form ionic bonds? (select all that apply.) chlorine (cl) calcium (ca) helium (he) sodium (na) fluorine (f) carbon (c), potassium is a _____.
One Has Been Shown For You.
It is a type of chemical bond that generates two oppositely charged ions. (b) list five examples of compounds with ionic bonds. Web chemistry chemistry questions and answers (a) use the periodic table to describe which elements form ionic bonds, and which elements form covalent bonds. Web between which type of elements do ionic bonds occur, and how do electrons act within the bond?
Such A Bond Forms When The Valence (Outermost) Electrons Of One Atom Are Transferred Permanently To Another Atom.
Metals and metals, electrons freely moving. Elements from opposite sides of the periodic table; Web ionic bonding is the complete transfer of valence electron(s) between atoms. Web ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound.
Metals And Nonmetals, Electrons Transferred.
One that tends to lose electrons and one that tends to gain them suppose an oxygen atom gains two electrons to become an oxygen ion. Web ionic bonding is the complete transfer of valence electron (s) between atoms and is a type of chemical bond that generates two oppositely charged ions. 3) last example, mg and cl. 1) for example, consider na and cl.
/ionic-bond-58fd4ea73df78ca1590682ad.jpg)

.PNG)