Can Nonpolar Molecules Form Hydrogen Bonds
Can Nonpolar Molecules Form Hydrogen Bonds - (i can't find a satisfying explanation) apparently, there's a misconception that says it's because of the. Web answer (1 of 3): Propane is nonpolar, because it is symmetric, with h atoms. Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; When they form a bond by contributing one electron each and then. As hydrogen molecule is made up of two hydrogen atoms having equal electronegativity. Covalent molecules made of only one type of atom, like. Web answer (1 of 8): 1 (a) conventional hydrogen bond compared with (b) a dihydrogen bond involving hydridic hydrogen atoms bonded to a strongly polarising element m. Examples include urea and polyurethane and the natural polymer.
Web because nonpolar molecules share their charges evenly, they do not react to electrostatic charges like water does. 1 (a) conventional hydrogen bond compared with (b) a dihydrogen bond involving hydridic hydrogen atoms bonded to a strongly polarising element m. When they form a bond by contributing one electron each and then. If there is an acceptable difference. The polarity of these molecules indicates that they will. Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar molecules). Web answer (1 of 8): It results from the attractive force between a. An example of a non polar molecule that is able to form hydrogen bonds is the amino acid tryptophan due its indole. Web distinguish between the following three types of intermolecular forces:
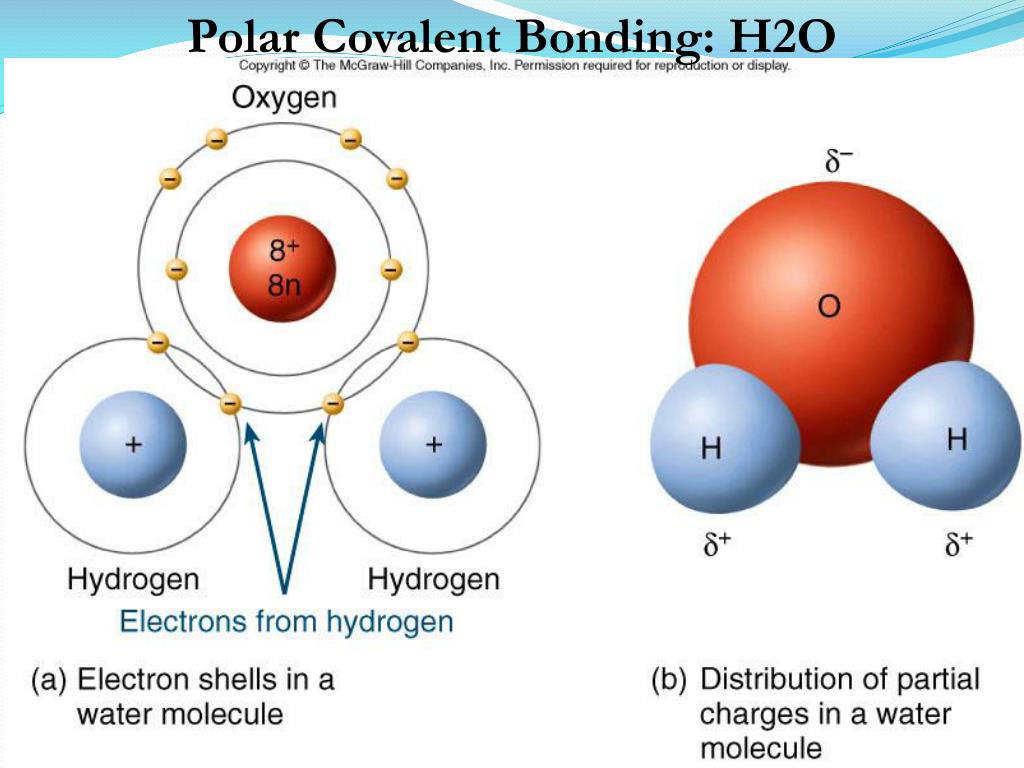
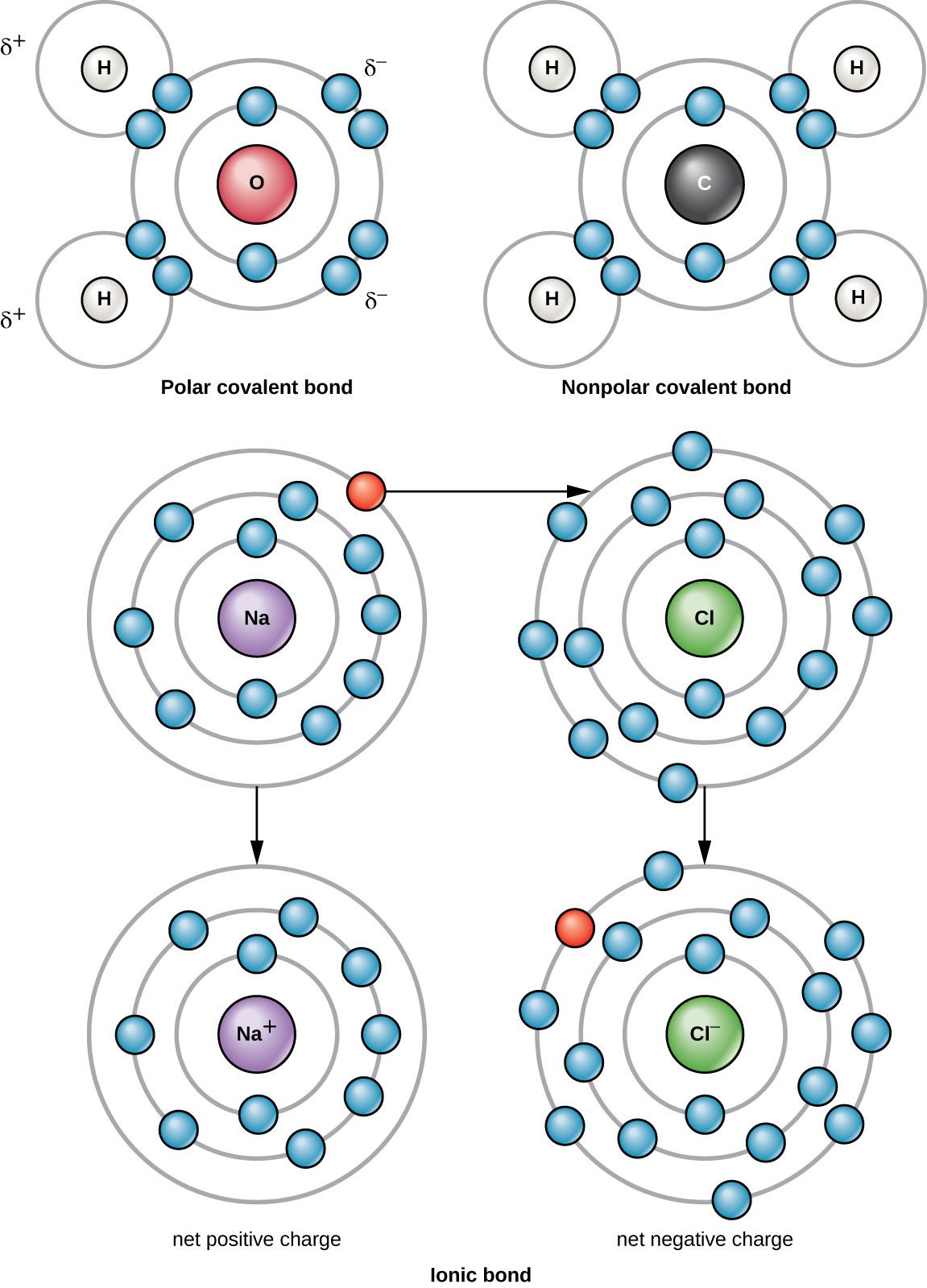
The two oxygen atoms pull on the electrons by exactly the same amount. Web the polarity of a covalent bond can be judged by determining the difference of the electronegativities of the two atoms involved in the covalent bond, as summarized. Examples include urea and polyurethane and the natural polymer. The hydrogen bond in polar molecules occurs only in compounds that have hydrogen bonded to n, o, or f. Most non polar molecules don't. The polarity of these molecules indicates that they will. Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar molecules). Web answer (1 of 8): Web water molecules form hydration shells around the ions: Positively charged na ^+ + ions are surrounded by partial negative charges from the oxygen ends of the water molecules,.
Ch4 Polar Or Nonpolar Atom Closest To Negative Side Is Carbon Dioxide
Web the molecule is symmetric. (i can't find a satisfying explanation) apparently, there's a misconception that says it's because of the. Positively charged na ^+ + ions are surrounded by partial negative charges from the oxygen ends of the water molecules,. Web the polarity of a covalent bond can be judged by determining the difference of the electronegativities of the.
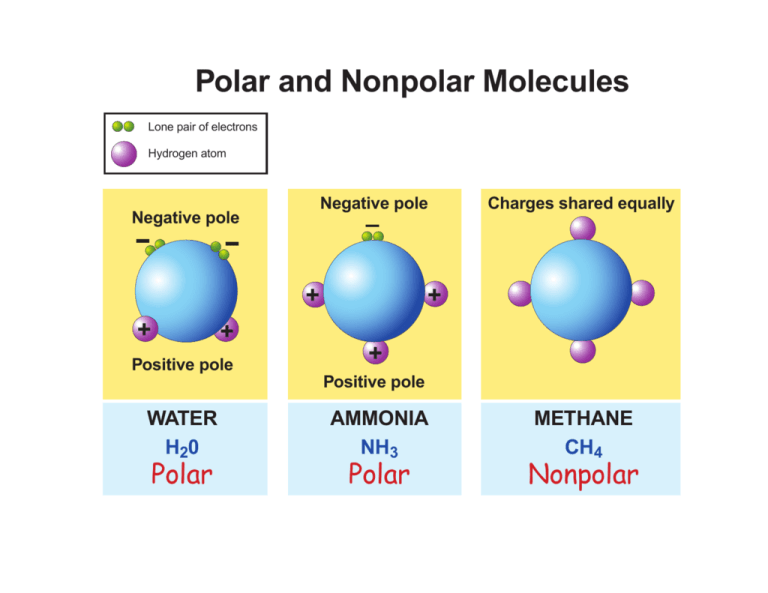
Polar and Nonpolar Molecules
Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar molecules). Identify types of intermolecular forces. Most non polar molecules don't. The two oxygen atoms pull on the electrons by exactly the same amount. Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar.
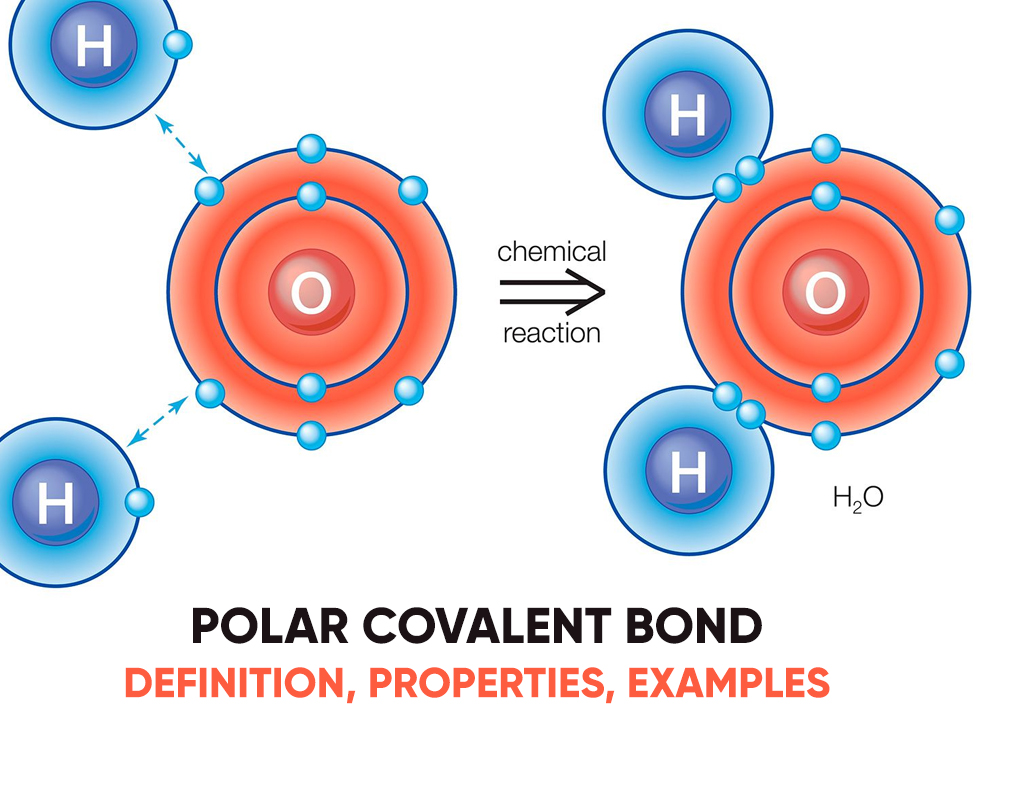
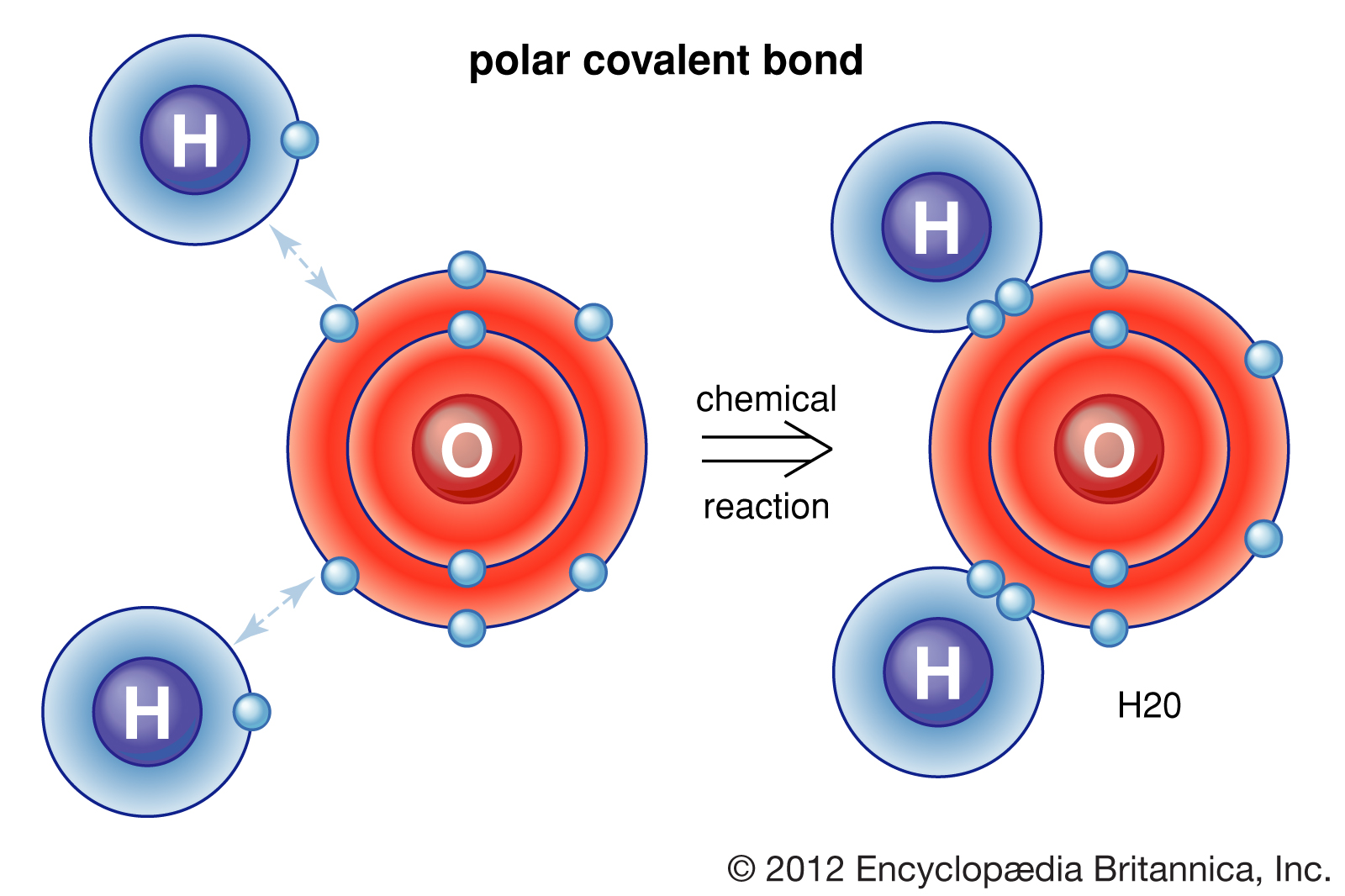
Polar Covalent Bond Definition, Properties, Examples
Polymers that contain carbonyl or amide groups can form hydrogen bonds. Web distinguish between the following three types of intermolecular forces: It results from the attractive force between a. Most non polar molecules don't. Web because nonpolar molecules share their charges evenly, they do not react to electrostatic charges like water does.
Ch4 Polar Or Nonpolar Covalent Bond PPT The Chemistry of Life
Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; Web because nonpolar molecules share their charges evenly, they do not react to electrostatic charges like water does. As hydrogen molecule is made up of two hydrogen atoms having equal electronegativity. Web whether atoms form polar or nonpolar bonds.
Bonds That Hold Water Molecules Together / Intermolecular Forces
Polymers that contain carbonyl or amide groups can form hydrogen bonds. 1 (a) conventional hydrogen bond compared with (b) a dihydrogen bond involving hydridic hydrogen atoms bonded to a strongly polarising element m. It results from the attractive force between a. Propane is nonpolar, because it is symmetric, with h atoms. Web water molecules form hydration shells around the ions:
2.2A Covalent Bonds and Other Bonds and Interactions Medicine LibreTexts
Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; The two oxygen atoms pull on the electrons by exactly the same amount. Such a bond is weaker than an ionic bond or. The hydrogen bond in polar molecules occurs only in compounds that have hydrogen bonded to n,.
Ch4 Polar Or Nonpolar Covalent Bond A CH4 B H2O C CF4 D CH3F Non
Web the molecule is symmetric. (i can't find a satisfying explanation) apparently, there's a misconception that says it's because of the. Propane is nonpolar, because it is symmetric, with h atoms. Web because nonpolar molecules share their charges evenly, they do not react to electrostatic charges like water does. Web this, without taking hydrogen bonds into account, is due to.
التربويون الجدد الفرق بين الروابط الأيونية والتساهمية The difference
The two oxygen atoms pull on the electrons by exactly the same amount. Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar molecules). The polarity of these molecules indicates that they will. If there is an acceptable difference. Identify types of intermolecular forces.
Chemical Bonds Anatomy and Physiology I
Web whether atoms form polar or nonpolar bonds depends on the difference between their electronegativity values. It results from the attractive force between a. Web answer (1 of 8): If there is an acceptable difference. An example of a non polar molecule that is able to form hydrogen bonds is the amino acid tryptophan due its indole.
Ch4 Polar Or Nonpolar Covalent Bond PPT The Chemistry of Life
Polymers that contain carbonyl or amide groups can form hydrogen bonds. The hydrogen bond in polar molecules occurs only in compounds that have hydrogen bonded to n, o, or f. Web water molecules form hydration shells around the ions: Identify types of intermolecular forces. Web the molecule is symmetric.
Such A Bond Is Weaker Than An Ionic Bond Or.
The hydrogen bond in polar molecules occurs only in compounds that have hydrogen bonded to n, o, or f. Positively charged na ^+ + ions are surrounded by partial negative charges from the oxygen ends of the water molecules,. As hydrogen molecule is made up of two hydrogen atoms having equal electronegativity. Web some examples of polar molecules which can hydrogen bond are ammonia (#nh_3#) and methanol (#ch_3oh#).
Most Non Polar Molecules Don't.
The two oxygen atoms pull on the electrons by exactly the same amount. Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar molecules). Propane is nonpolar, because it is symmetric, with h atoms. The polarity of these molecules indicates that they will.
Covalent Molecules Made Of Only One Type Of Atom, Like.
(i can't find a satisfying explanation) apparently, there's a misconception that says it's because of the. It results from the attractive force between a. Web the polarity of a covalent bond can be judged by determining the difference of the electronegativities of the two atoms involved in the covalent bond, as summarized. Web this, without taking hydrogen bonds into account, is due to greater dispersion forces (see interactions between nonpolar molecules).
1 (A) Conventional Hydrogen Bond Compared With (B) A Dihydrogen Bond Involving Hydridic Hydrogen Atoms Bonded To A Strongly Polarising Element M.
Web because nonpolar molecules share their charges evenly, they do not react to electrostatic charges like water does. Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; Web answer (1 of 3): Examples include urea and polyurethane and the natural polymer.