Acids React With Bases To Form Salt And Water
Acids React With Bases To Form Salt And Water - You are missing a couple of symbols: Salts are odourless and have a salty taste, and many are soluble in water. Web a base is any substance that reacts with an acid to form a salt and water only. As discussed previously, metals that are more active than. A oxidation reaction b neutralisation reaction c reduction reaction d ionisation reaction. Acid + base → salt + water. Bases that are soluble in. Web answer (1 of 4): Acid + base → salt + water. Web question a base reacts with an acid to form salt and water.
Web acid + base = salt + water when acids react with bases, they produce a salt and water. Metal oxides are bases, because they neutralise acids. Acid + base → salt + water. H 2 so 4 (aq) + cuo(s) →. Sulfuric acid + copper(ii) oxide → copper(ii) sulfate + water. Web when an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. Web a neutralization reaction can be defined as a chemical reaction in which an acid and base quantitatively react together to form a salt and water as products. A oxidation reaction b neutralisation reaction c reduction reaction d ionisation reaction. Web an acidic solution and a basic solution react together in a neutralization reaction that also forms a salt. Web neutralisation is a reaction between an acid and an alkali that forms a salt and water.
Web acids react with bases to form a salt and water. Salts are ionic compounds containing a positive ion other than h + and a negative ion. For example, sodium hydroxide and hydrochloric acid react to form sodium. This means that metal oxides and metal hydroxides are bases. When a seed (acid) played her bass (base) drum she became very upset and began to. Web when an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. Web a base is any substance that reacts with an acid to form a salt and water only. H 2 so 4 (aq) + cuo(s) →. Acid + base → salt + water this is perhaps one of the most famous word equations [1] that can help remember chemical. Web question a base reacts with an acid to form salt and water.
May 2020 Scienceandsf A Blog Published by Robert A. Lawler
Sulfuric acid + copper(ii) oxide → copper(ii) sulfate + water. Web when an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. Web the reactions between acids and bases to form salts and water are double displacement reactions. Web acids and bases react with metals. Acid + base → salt.
Lecture 19.1a Acid/Base Properties
Web when an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. Web a base is any substance that reacts with an acid to form a salt and water only. A oxidation reaction b neutralisation reaction c reduction reaction d ionisation reaction. H 2 so 4 (aq) + cuo(s) →..
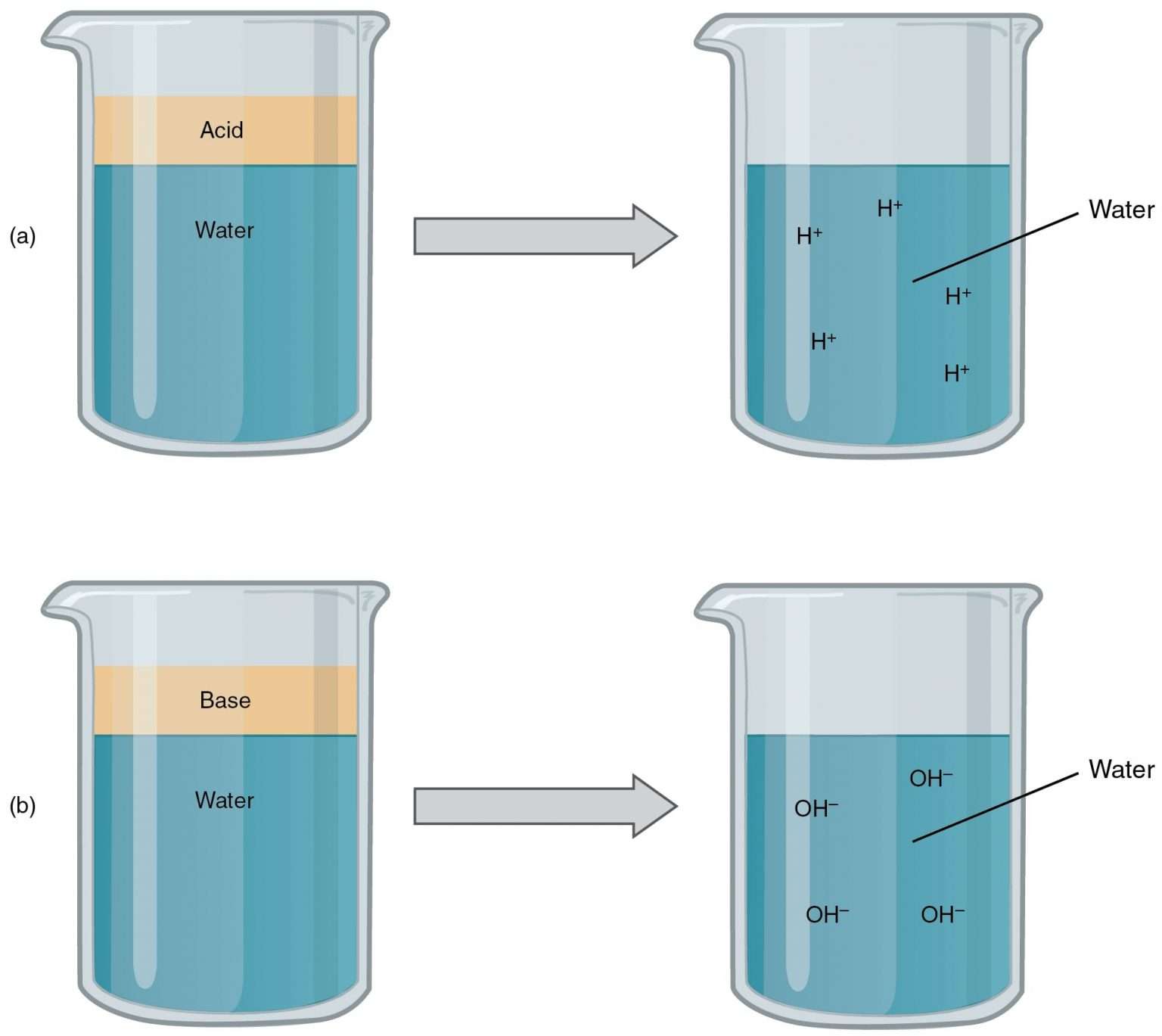
Acids Bases and Salts What happens when water is added to acid or
Web a base is any substance that reacts with an acid to form a salt and water only. Web a salt and water are produced when acids react with metal oxides. Web answer (1 of 4): For example, sodium hydroxide and hydrochloric acid react to form sodium. You are missing a couple of symbols:
Acid Bases and Salts Class 10 Science CBSE Chemical Properties of
Acid + base → salt + water this is perhaps one of the most famous word equations [1] that can help remember chemical. This means that metal oxides and metal hydroxides are bases. H 2 so 4 (aq) + cuo(s) →. Web answer (1 of 4): Web solution verified by toppr correct option is a) when acids react with base,.
Acids and Bases
As discussed previously, metals that are more active than. Web when an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. When a seed (acid) played her bass (base) drum she became very upset and began to. Bases that are soluble in. Acid + base → salt + water this.
Acids, bases and salts CPD RSC Education
Acid + base → salt + water this is perhaps one of the most famous word equations [1] that can help remember chemical. When a seed (acid) played her bass (base) drum she became very upset and began to. Nitric acid + magnesium oxide → magnesium nitrate + water. Web neutralisation is a reaction between an acid and an alkali.
Acids react with bases to form salt and water. This reaction is known as
Acid + metal oxide → salt + water. Web answer (1 of 4): Web when an acid and a base are placed together, they react to neutralize the acid and base properties, producing a salt. Salts are ionic compounds containing a positive ion other than h + and a negative ion. Web question a base reacts with an acid to.
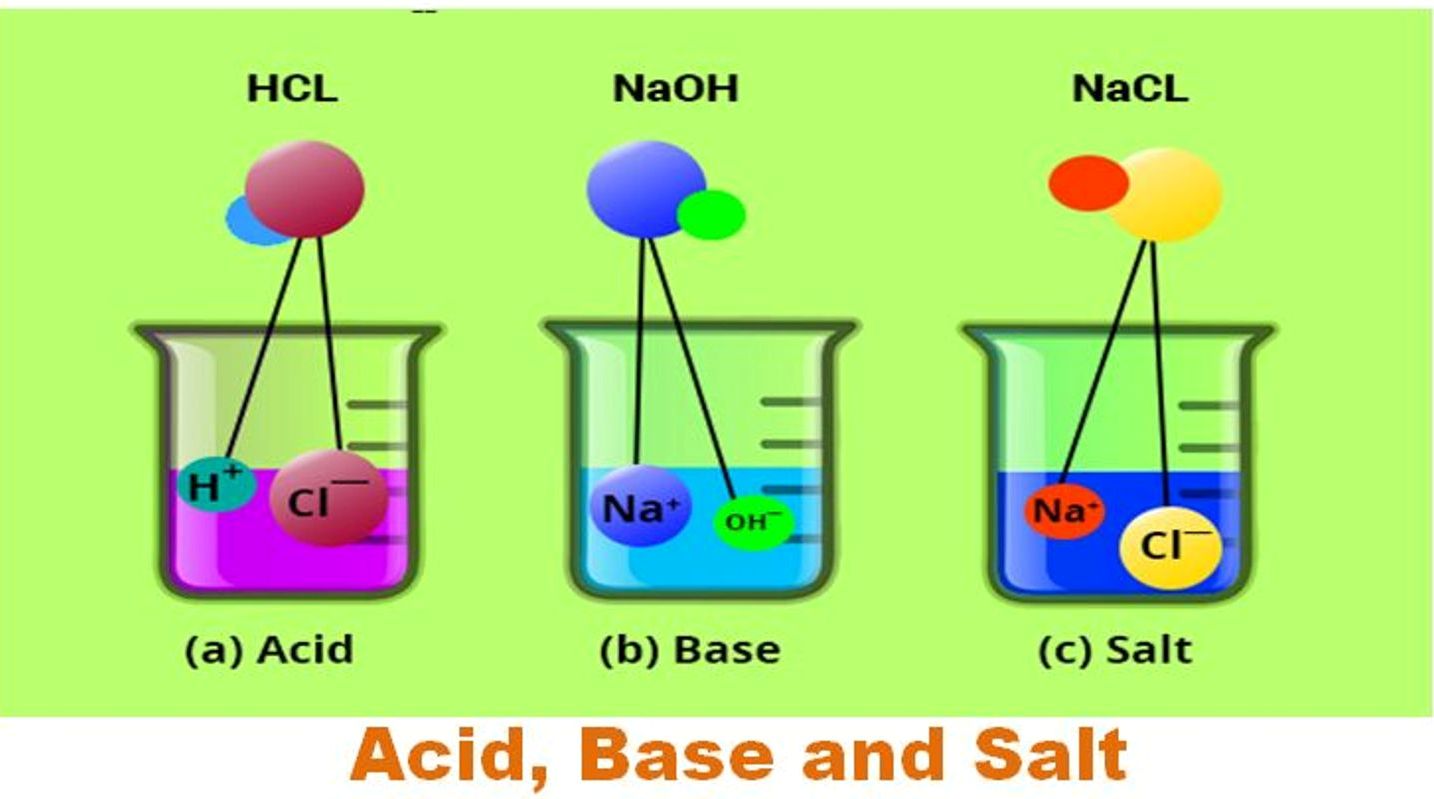
CBSE Chemistry 7th Acids bases and Salts [Solved] CBSE ADDA
Web a base is any substance that reacts with an acid to form a salt and water only. Web the reactions between acids and bases to form salts and water are double displacement reactions. Nitric acid + magnesium oxide → magnesium nitrate + water. Metal oxides are bases, because they neutralise acids. Web an acidic solution and a basic solution.
Acids and Bases 9 Properties, Useful Reaction & Examples
Web the reactions between acids and bases to form salts and water are double displacement reactions. Salts are ionic compounds containing a positive ion other than h + and a negative ion. A oxidation reaction b neutralisation reaction c reduction reaction d ionisation reaction. Web acids and bases react with metals. When acids react with a base, a salt and.
Chapter 8 Reactions in Aqueous Solution
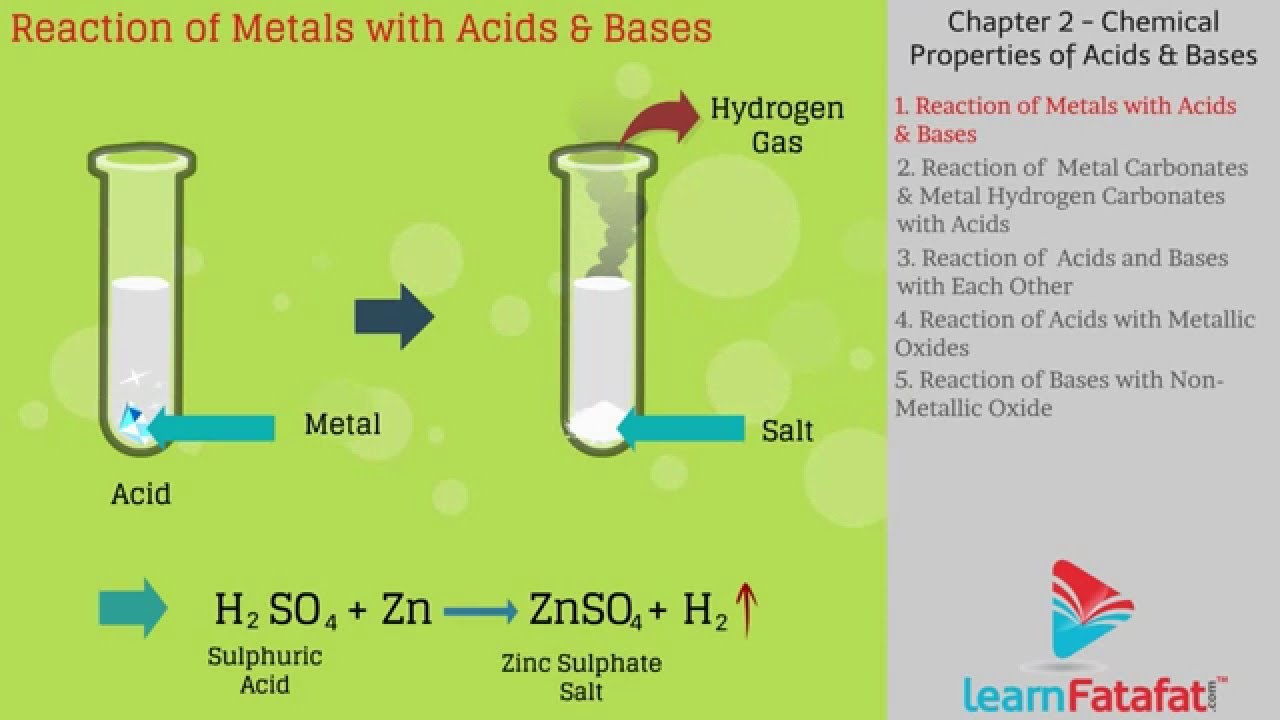
Web acid + base = salt + water when acids react with bases, they produce a salt and water. Web question a base reacts with an acid to form salt and water. Acid + base → salt + water. Acids react with most metals to form a salt and hydrogen gas. Web the reactions between acids and bases to form.
You Are Missing A Couple Of Symbols:
Nitric acid + magnesium oxide → magnesium nitrate + water. Acid + base → salt + water. Web answer (1 of 4): Sulfuric acid + copper(ii) oxide → copper(ii) sulfate + water.
Web When An Acid And A Base Are Placed Together, They React To Neutralize The Acid And Base Properties, Producing A Salt.
When a seed (acid) played her bass (base) drum she became very upset and began to. A oxidation reaction b neutralisation reaction c reduction reaction d ionisation reaction. Web solution verified by toppr correct option is a) when acids react with base, they neutralize each other's effect and thus it is called as neutralization reaction, energy is released. Bases that are soluble in.
Web A Base Is Any Substance That Reacts With An Acid To Form A Salt And Water Only.
This means that metal oxides and metal hydroxides are bases. Salts are ionic compounds containing a positive ion other than h + and a negative ion. Acid + base → salt + water this is perhaps one of the most famous word equations [1] that can help remember chemical. Web a salt and water are produced when acids react with metal oxides.
Web Acids And Bases React With Metals.
Salts are odourless and have a salty taste, and many are soluble in water. Web acids react with bases to form a salt and water. Acid + base → salt + water. Web question a base reacts with an acid to form salt and water.





![CBSE Chemistry 7th Acids bases and Salts [Solved] CBSE ADDA](https://1.bp.blogspot.com/-F3SKPLqiR9g/UZ5HGgkzuiI/AAAAAAAABZU/tCoRRXbdkgs/s1600/aCIDS+bASE+AND+sALTS.png)